A Look Into Biomarkers and How They Could Be Applied to STXBP1
Asish Madarapu
What are Biomarkers?
To start, biomarker is short for biological marker. The FDA defines a biomarker as a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacological responses to therapeutic interventions. What this means is that biomarkers refer to measurable indications of the medical state of a patient, i.e they show how a person is doing through measuring something. Biomarkers are not to be confused with symptoms though. Symptoms are indications of illness that can be perceived by the patient while biomarkers cannot be felt. For example, levels of cholesterol can be a biomarker for heart disease but the patient will not feel that they have high levels. Biomarkers can be a molecule, like cholesterol, a structure, like an X-ray, or process, like blood pressure.
Classifications of Biomarkers
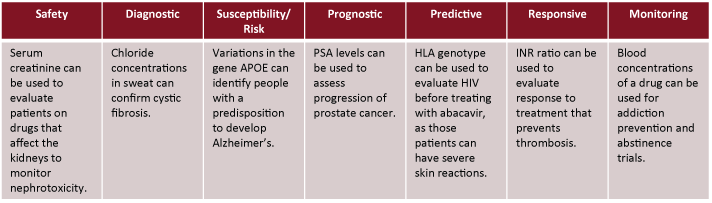
The FDA classifies biomarkers into 7 different categories: safety, diagnostic, susceptibility/risk, prognostic, predictive, responsive and monitoring(Ref 1). Here are brief descriptions for each:
Safety: Used to measure the presence or likelihood of toxicity as an adverse effect of exposure to medical products.
Diagnostic: Biomarker which confirms if a disease or condition is present.
Susceptibility/risk: Biomarker which indicates potential for developing a disease.
Prognostic: Biomarker used to identify recurrence of a disease or progression in a disease.
Predictive: Identifies individuals who are more likely than others to respond to a medical product, either positively or negatively.
Responsive: Biomarker which shows biological response to medical products and/or environmental agents.
Monitoring: Biomarker used to measure the effects of a treatment and the body's response to it. This is measured numerous times to record progress.
Categories of Biomarkers
The table below lists some examples of biomarkers:
How are biomarkers used?
Biomarkers play an important role in medicine and drug development. How are biomarkers used in medicine? Well many can be used to monitor risk to organs in the body. Take blood pressure for example. Blood pressure is measured commonly for cardiovascular health, as it can track risk for heart attack/strokes. Additionally it can see how well medication is working like an ACE inhibitor. This relaxes blood vessels, and in turn lowers blood pressure.
How are biomarkers used in drug development? Drugs are evaluated in clinical trials, and using biomarkers can help in this by performing 4 important roles.
Biomarkers can help identify individuals for trials who may respond best to the drug being tested. Identifying individuals for a trial is quite important for ensuring success or a drug or medication. Populations can be identified that are more likely to have a favorable outcome and/or have decreased toxicity (Ref 3).
Biomarkers can be used to help determine if a drug is interacting at its intended biological target in the body. Drugs work by interacting with molecules in our cells and tissues. This interaction may cause an increase or decrease in the amount of other molecules, which could be used to help show that the drug is working correctly.
Biomarkers are used to tell if a drug is having an unintended toxic, or adverse effect on the body. Safety monitoring is a key component of clinical trials.
Biomarkers can help monitor a patient’s responses to the drug to see if the drug is having its intended beneficial effect (Ref 2). This is the goal of a clinical trial, to see if a drug is effective in treating a disease or disorder.
This last use of biomarkers for drug development can be very important. The problem now with drug development is how many clinical trials are unsuccessful. Getting past the clinical process takes long amounts of time and costs a lot as well. Biomarkers can come into play here. Biomarkers can early on, indicate effectiveness of a drug. Surrogate endpoints are biomarkers that can be used to validate a clinical trial. Surrogate endpoints can substitute for a medical or clinical endpoint, which are the types of endpoints usually used to determine if a drug is effective in a clinical trial. For example, a drug may increase survival in patients, which is a clinical endpoint, but this takes a long time to measure accurately. A surrogate endpoint might be used and measure effectiveness in a shorter time frame. An example of a surrogate endpoint is tumor size in various cancers. If this is used in clinical trials, and there is reduction in tumor size in response to a drug, this can prove the effectiveness of a medicine rather than waiting to see if survival rates are increased, which could take years to prove.
Using surrogate endpoints, the research regarding new medicines can be accelerated. Information can be obtained faster and at a lower cost (Ref 4). New treatments can be provided faster for patients as more biomarkers are being used and discovered in research. Surrogate endpoints do come with disadvantages though. Relationships between a specific biomarker and disease progression may not be uniform, so identifying a sufficient measure can prove hard. Many diseases arise from a combination of physiological defects and can hinder the ability to find an uncomplicated surrogate endpoint. Some relationships may be misinterpreted and this could be risky for patients' health. Thus, they should be approached in a careful manner.
Potential Applications of Biomarkers for STXBP1
STXBP1 is a genetic disorder, and no two people with the disease have identical symptoms. As a result, drug development is quite tricky when approaching diseases like STXBP1. This is where biomarkers can help. Certain categories of biomarkers can be used for STXBP1.
Diagnostic: STXBP1 disorder is caused by a mutation in the syntaxin binding protein 1 gene. Identifying that a person has a mutation in the gene is sufficient to diagnose the disorder.
Susceptibility/risk: Identifying the type of STXBP1 mutation in a patient may help identify the risk of developing certain symptoms, or in cases of mosaicism, identifying the percentage of cells that have a mutation might be used to help determine the extent of symptoms.
Predictive: This biomarker category could prove very useful. STXBP1 patients have varying degrees of symptoms, e.g. some patients have seizures and others do not. Subsets of STXBP1 patients could be chosen who might better respond to certain treatments using this type of biomarker.
Safety: Assessing safety of a drug is very important. Many STXBP1 patients are nonverbal, thus might not be able to tell their caregiver if a drug is causing them distress. Biomarkers can be used to indicate negative effects to a treatment.
Responsive/Monitoring: Because STXBP1 is a neurodevelopmental disorder, it could take a long time for a drug to change brain function enough before it becomes apparent. If a biomarker could be identified that changes more quickly, it could be used to indicate a particular drug is working and should be continued to be used by the patient. These types of biomarkers can show the body’s response to a treatment which is essential in validating a drug.
When thinking about developing biomarkers for STXBP1, there are some ideal characteristics that should be considered.
The biomarker should be significantly different in STXBP1 patients compared to other neurotypical control populations.
The biomarker should be quantifiable, i.e. measurable, in an accessible manner. Meaning that it should be easy to perform, like a blood test is easy to perform.
The biomarker should be relatively quick and straightforward to measure.
The biomarker should reflect a clinical outcome. That means that a positive change in the biomarker is associated with a positive change in patient symptoms or well being.
One potential biomarker for STXBP1 that fits all the above criteria is electroencephalography, or EEG. A study published in December of 2021 compared EEGs from STXBP1 children to EEGs from neurotypical children. The main finding of this paper was that there was a greater ‘global inhibitory tone’ in the EEGs from STXBP1 patients as measured by a decrease in excitatory/inhibitory ratio (fE/I). See the figure below. EEGs are one of the many options that can be used as a biomarker for approaching treatments to STXBP1. EEGs could be used as a monitoring, prognostic, predictive, and/or response biomarker. This provides numerous possibilities. Identifying biomarkers like these can bring a personalized treatment for each patient to ensure the most efficient way of dealing with STXBP1.
EEGs from STXBP1 children appear to show a greater inhibitory tone as demonstrated by a decrease in excitatory/inhibitory balance (fE/I). From Ref 5.
In summary, biomarkers are an important tool in medicine and drug development. The identification and use of biomarkers for STXBP1 will be important as we move towards generating therapies.
Sources/References
BEST (Biomarkers, EndpointS, and other Tools) Resource https://www.ncbi.nlm.nih.gov/books/NBK338448/
Selleck MJ et al., (2017). Making Meaningful Clinical Use of Biomarkers. Biomark Insights, 12:1.
Mandreker S.J. and Sargent DJ (2009). Clinical Trial Designs for Predictive Biomarker Validation: One Size Does Not Fit All. J Biopharm Stat, 19(3):530.
Aronson JK (2005). Biomarkers and Surrogate Endpoints. Br J Clin Pharmacol, 59(5):491.
Houtman SJ et al., (2021). STXBP1 Syndrome is Characterized by Inhibition-Dominated Dynamics of Resting-State EEG. Front Physiol, 23:Article 775172.